Production of microalgae biofuel while reducing wastes.
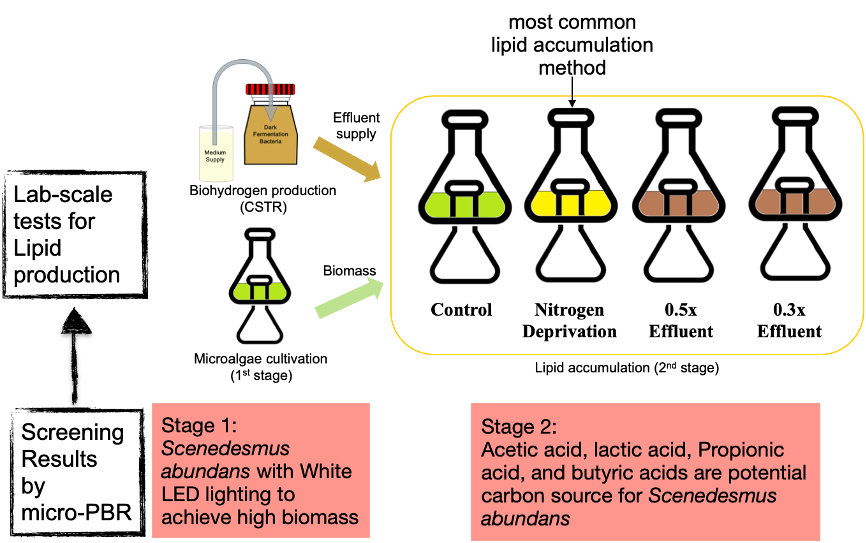
Microalgae can produce abundant cellular lipids that can be transformed into biodiesel to reduce the demand of fossil fuels. High microalgal lipid yields can be obtained under multiple combinations of cultivation and stress strategies such as heterotrophic culture, mixotrophic culture, high salt, high light, nitrogen depletion, and phosphorus depletion. These strategies have their pros and cons and are widely applied in increasing the production of biofuel. We applied the effluent from biohydrogen production on the induction of lipid accumulation in S. abundans and obtained the highest values of biomass and lipid yields, 2.831 g/L and 1.238 g/L respectively, which are the highest among existing reports. S. abundans also had extremely high removal efficiencies for organic acids in the biohydrogen effluent (formic acid: 100%, acetic acid: 100%, propionic acid: 98.35%, lactic acid: 84.01%, and butyric acid: 67.69%). These results indicate that S. abundans has great abilities in dealing with the effluent from the dark anaerobic fermentation of biohydrogen production and transforming the organic acids into biomass and cellular lipids. This is beneficial in the future integration of biohydrogen and microalgal lipid productions to reduce the potential pollution by organic acids and improve the energy efficiency of bioenergy.Paragraph. 按此處以編輯.
Production of electricity from wastewater using microbial fuel cells.
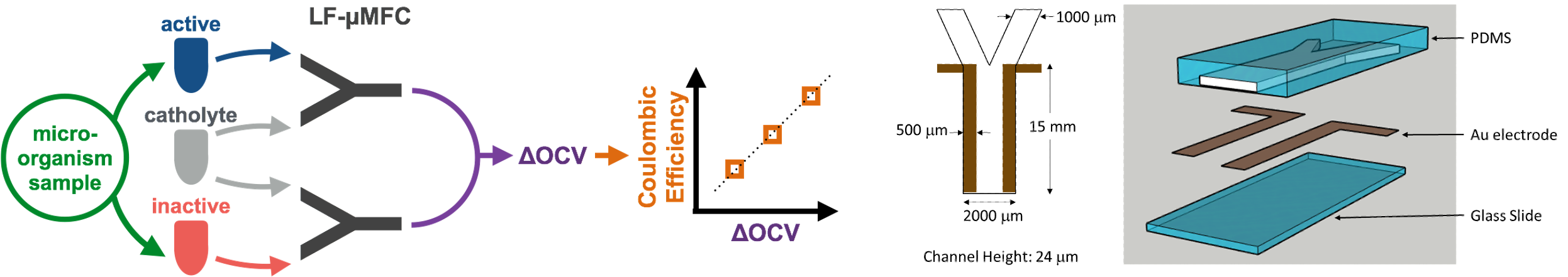
To our best knowledge, we have establish the first technology capable of achieving quantitative evaluation of performance of larger scale microbial fuel cells (MFCs) based on the results from the microfluidic assay, and probably the only rapid quantification method for measuring the Coulombic efficiency of the microbial electrochemical systems. Our quantification method applies principles that are new to conventional methods and current microfluidic methods. The rapid quantification is accomplished by a microfluidic electroactivity analysis which measures the open voltage difference (ΔOCV) between active and inactivated microorganism samples. This technology obtained highly linear correlations between ΔOCV from the microfluidic assay and Coulombic efficiency from the lab-scale H-type MFC with 3 pure strains microorganisms (Shewanella oneidensis MR-1, Aeromonas hydrophila NIU01, and Proteus hauseri ZMd44) fed with 4 carbon sources (acetate, glucose, glycerol, sucrose). The linear calibration was also applied to predict the Coulombic efficiency of the lab-scale H-type MFC with mixed culture microorganisms. The prediction was highly accurate with a maximum error of 0.053%. These results show that our microfluidic electroactivity assay is an exceedingly effective tool for screening microorganisms and carbons sources for efficient electron recovery.
