Our research topics in microfluidics focus on the controlling of fluid behavior and cell behavior. For example, the two-phase flow inside a microfluidic device can be co-flow (and laminar), turbulent (mixing), or forming uniform droplet. Following is one example of controlling the formation of polymer droplets inside microfluidic device.
Applications include:
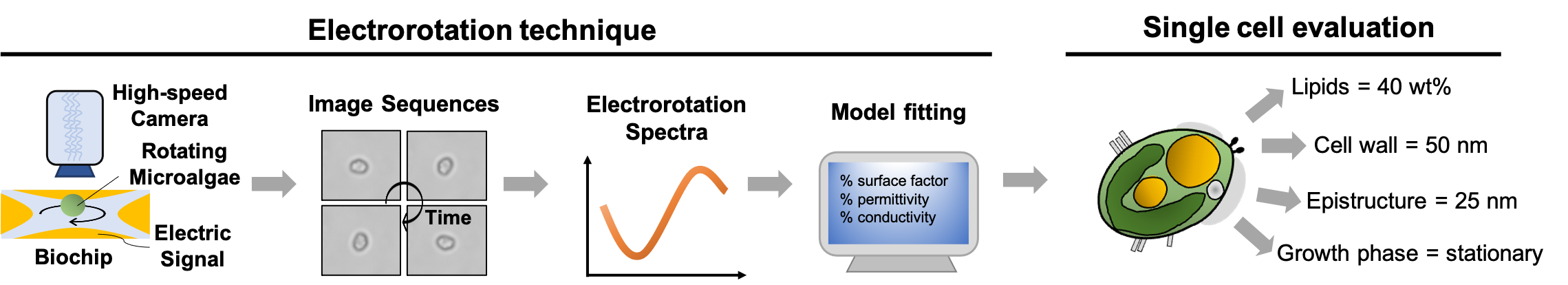
Developing alternative energy is a common interest around the world due to the shortage of fossil fuels. Using microalgae to produce lipids, which can be further transesterificated to biodiesel, has become a favorable choice because the production is a carbon-neutral process and it requires small land space and short cultivation cycles. The conventional methods for quantifying cellular lipids inside microalgae can be divided into two categories: one involves several physical or chemical processes for purifying the microalgal oil and the other relies on the fluorescence labeling of the cellular lipids. The methods in the first category are time and labor consuming while the latter is rapid and straightforward. However, the fluorescence method has been reported to produce inconsistent results. The variations in cell sizes, structures as well as the compositions of cell membrane are considered as possible reasons for these inconsistent results. Large cell size, stiff cell membrane, and inserting of probes into the lipid bilayer are not favorable for the probe to reach the cellular lipid droplets.
We have developed 2 noninvasive and rapid quantification methods for microalgae cellular contents, single cell electrorotation and near-infrared quantitative Raman spectroscopy, to overcome the shortage of current lipid quantification methods. The real-time information can serve as feedback for improving the cultivation conditions and as basis for microalgae strain screening.
In the publication of “Electrorotation of single microalgae cells during lipid accumulation for assessing cellular dielectric properties and total lipid contents” (Biosensors and Bioelectronics 2021), we demonstrated the electrorotation of single microalgae cells and apply the technology to accurately quantify cellular lipids. This is a collaborative research project with Prof. Le Pioufle in ENS-Cachan and my Ph.D. student, Yu-Sheng Lin, has conducted the major part of the experiment and simulation. Prof. Le Pioufle provides knowledge and facility to conduct the electrorotation experiment while the applicant provides knowledge and experiment design for the microalgae cultivation and lipid induction. Related research outcomes are also published in other renowned international conferences including “The 22nd International Conference on Miniaturized Systems for Chemistry and Life Sciences (µTAS 2018)” and “The 25th symposium of Young Asian Biological Engineers' Community (YABEC 2019)”.
Applications include:
- Develop microfluidic analysis technology for manipulating cells such as dielectrophoresis and electrorotation.
- Develop microfluidic systems that mimics biological environment for the purpose of in vitro drug screening platforms or synthetic organs.
- Develop microfluidic systems to find optimized parameters for large scale bio-processes such as heavy metal removal from water and microplastic removal from the ocean.
Developing alternative energy is a common interest around the world due to the shortage of fossil fuels. Using microalgae to produce lipids, which can be further transesterificated to biodiesel, has become a favorable choice because the production is a carbon-neutral process and it requires small land space and short cultivation cycles. The conventional methods for quantifying cellular lipids inside microalgae can be divided into two categories: one involves several physical or chemical processes for purifying the microalgal oil and the other relies on the fluorescence labeling of the cellular lipids. The methods in the first category are time and labor consuming while the latter is rapid and straightforward. However, the fluorescence method has been reported to produce inconsistent results. The variations in cell sizes, structures as well as the compositions of cell membrane are considered as possible reasons for these inconsistent results. Large cell size, stiff cell membrane, and inserting of probes into the lipid bilayer are not favorable for the probe to reach the cellular lipid droplets.
We have developed 2 noninvasive and rapid quantification methods for microalgae cellular contents, single cell electrorotation and near-infrared quantitative Raman spectroscopy, to overcome the shortage of current lipid quantification methods. The real-time information can serve as feedback for improving the cultivation conditions and as basis for microalgae strain screening.
In the publication of “Electrorotation of single microalgae cells during lipid accumulation for assessing cellular dielectric properties and total lipid contents” (Biosensors and Bioelectronics 2021), we demonstrated the electrorotation of single microalgae cells and apply the technology to accurately quantify cellular lipids. This is a collaborative research project with Prof. Le Pioufle in ENS-Cachan and my Ph.D. student, Yu-Sheng Lin, has conducted the major part of the experiment and simulation. Prof. Le Pioufle provides knowledge and facility to conduct the electrorotation experiment while the applicant provides knowledge and experiment design for the microalgae cultivation and lipid induction. Related research outcomes are also published in other renowned international conferences including “The 22nd International Conference on Miniaturized Systems for Chemistry and Life Sciences (µTAS 2018)” and “The 25th symposium of Young Asian Biological Engineers' Community (YABEC 2019)”.
More applications will be updated soon.
